A practical, evidence-based, physiology-first ED approach — and the pitfalls to avoid
In the emergency department, PE is one of the classic traps.
Miss it, and the patient can deteriorate fast.
Overcall it, and you expose people to unnecessary CT, contrast, radiation, false positives, and treatment they never needed.
The 2026 AHA/ACC multisociety guideline pushes a very important message:
PE is not a “CT first” diagnosis. It is a probability-first diagnosis.
That is the right ER mindset.
The first principle: before you diagnose PE, understand what PE does
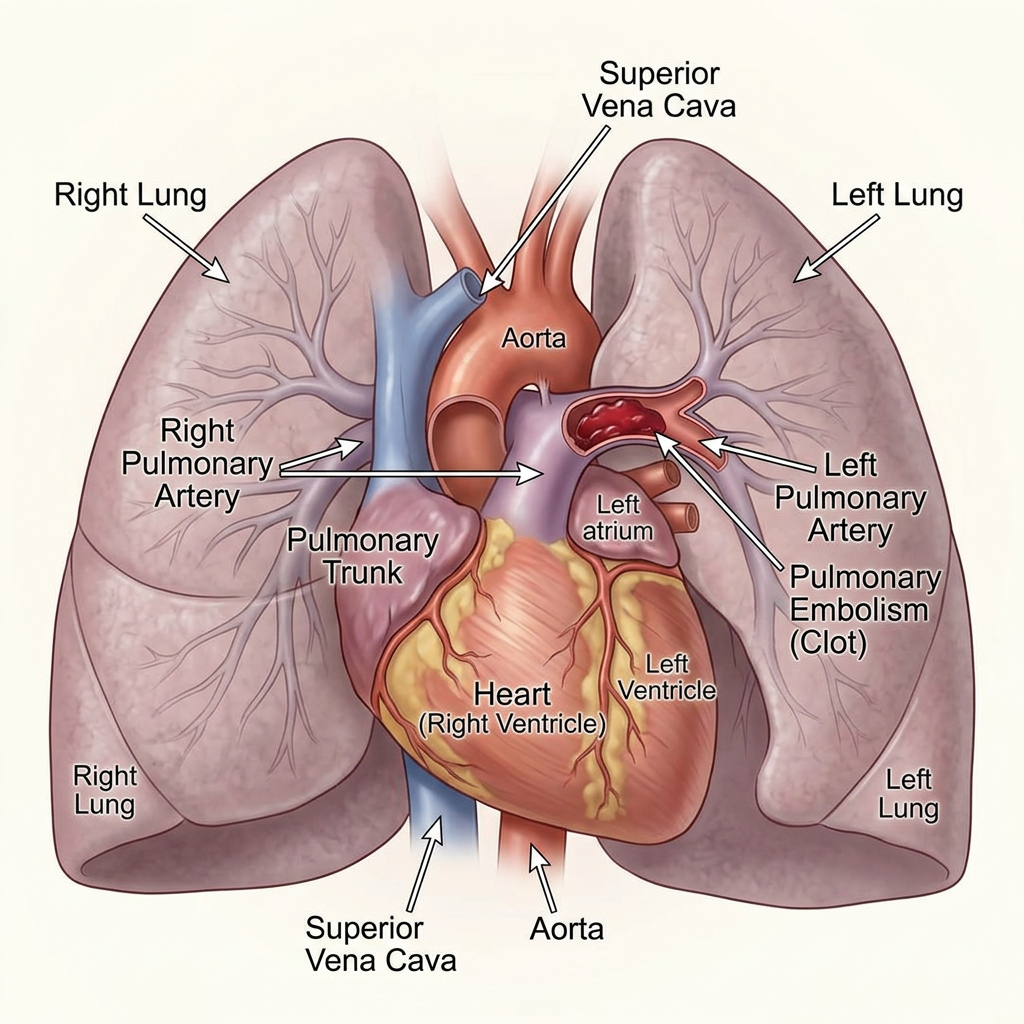
A pulmonary embolus is not just “a clot in the lung.”
Physiologically, it does 3 main things:
1. It blocks pulmonary blood flow.
That increases pulmonary vascular resistance and suddenly raises RV afterload.
2. It creates dead-space physiology.
Parts of the lung are ventilated but not perfused, so gas exchange becomes inefficient. The patient becomes tachypneic, dyspneic, and sometimes hypoxemic.
3. In more severe cases, it becomes an RV disease.
The RV is a thin-walled chamber built for volume, not pressure. When afterload rises abruptly, the RV dilates, wall stress rises, coronary perfusion worsens, the septum shifts leftward, LV filling falls, cardiac output drops, and the patient can spiral into shock.
That physiology explains the whole bedside spectrum:
- pleuritic pain and dyspnea in smaller PE
- tachycardia and hypoxemia in moderate PE
- syncope, hypotension, lactate elevation, RV strain, and collapse in severe PE
So when I evaluate PE, I ask 2 questions at the same time:
1-Could this be PE?
2- Is this clot burden now causing meaningful cardiopulmonary stress?
That second question is where good emergency medicine matters.
Step 1: Decide whether this is a PE workup at all
Not every dyspneic or tachycardic patient needs a PE pathway.
Start with a real bedside assessment:
- sudden unexplained dyspnea
- pleuritic chest pain
- syncope
- unexplained tachycardia
- unexplained hypoxemia
- signs or symptoms of DVT
- recent surgery, immobility, travel, cancer, estrogen exposure, pregnancy/postpartum state, prior VTE
But do not stop thinking broadly. The patient may have:
- pneumonia
- ACS
- heart failure
- COPD/asthma
- pneumothorax
- pericardial tamponade
- sepsis
- anemia
- metabolic acidosis
A good ED physician does not ask, “Can I order a D-dimer?”
He asks, “Does this patient actually earn a PE workup?”
That single discipline prevents a lot of bad medicine.
Step 2: First separate the sick patient from the stable patient
Before scores, before algorithms, before D-dimer:
Is the patient unstable?
Look at:
- blood pressure
- MAP
- mental status
- lactate
- work of breathing
- oxygen requirement
- signs of RV strain or shock
- whether this feels like impending decompensation
This matters because the stable and unstable PE workups are not the same.
If unstable
In the crashing patient, PE becomes a resuscitation diagnosis, not just a radiology diagnosis.
You may not have the luxury of a perfect outpatient-style algorithm. In that patient, bedside echo, shock physiology, ECG clues, ultrasound evidence of DVT, and overall context become much more important. The guideline is clear that echocardiography alone should not be used to confirm or exclude PE in routine diagnosis, but in hemodynamic compromise it becomes a critical tool for rapid physiologic assessment and treatment direction.
In plain ER language:
In stable patients, PE diagnosis is a probability pathway.
In unstable patients, PE diagnosis becomes a physiology-and-time pathway.
Also, the guideline emphasizes that if suspected PE is serious enough and imaging is delayed or not immediately accessible, therapeutic anticoagulation may be beneficial in patients with higher-risk suspected PE and low bleeding risk.
Step 3: Estimate pretest probability before ordering tests
This is the center of the whole strategy.
Use:
- Wells
- revised Geneva
- or honest clinical gestalt
The guideline allows all three. What matters is not which score makes you feel sophisticated. What matters is that you categorize the patient before testing.
I simplify it into 3 buckets:
- Low probability
- Intermediate probability
- High probability
Once you know the bucket, the next step becomes obvious.
Step 4: In low-risk patients, ask whether you can stop with PERC
If the patient is truly low risk, use PERC.
This is where many clinicians go wrong.
PERC is not a PE test.
PERC is a test-avoidance tool.
It is only for patients who are already low probability.
If all PERC criteria are negative, the post-test probability becomes low enough that no D-dimer and no imaging are needed.
That is powerful. It avoids unnecessary cascades.
Practical PERC message
Use PERC only when your honest bedside impression is:
“PE is possible, but already unlikely.”
Do not use PERC to talk yourself out of a PE you are actually worried about.
That is one of the most common cognitive errors in the ED.
Step 5: In low-to-intermediate risk patients, use D-dimer properly
D-dimer is useful, but only when you respect what it is.
It is a marker of fibrin formation and breakdown.
So it tells you that clot turnover is happening somewhere.
That gives it good sensitivity for PE.
But poor specificity.
D-dimer goes up in:
- infection
- inflammation
- recent surgery
- cancer
- trauma
- pregnancy
- aging
- hospitalization
- many other inflammatory states
So D-dimer is a rule-out test, not a rule-in test.
When it helps
It helps in:
- low-risk patients who are not PERC negative
- intermediate-risk patients
When it does not help
It does not help when the patient is high probability.
Once pretest probability is high, you should stop asking a screening blood test to rescue the situation. That patient needs imaging.
Step 6: Use age-adjusted D-dimer or YEARS to reduce unnecessary CT
This is one of the most practical updates from contemporary PE diagnosis.
Age-adjusted D-dimer
In adults with low or intermediate clinical probability, an age-adjusted D-dimer can safely exclude PE.
Why this makes physiologic sense:
older patients often have higher baseline fibrin turnover and more false-positive D-dimers. Using a fixed threshold in all older adults floods the system with unnecessary scans.
This is one of the cleanest examples of smarter testing rather than more testing.
YEARS
The guideline supports the YEARS algorithm as another useful imaging-sparing strategy.
YEARS asks 3 questions:
- Are there clinical signs of DVT?
- Is there hemoptysis?
- Is PE the most likely diagnosis?
Then D-dimer is interpreted in context:
- 0 YEARS items → a higher D-dimer threshold can still exclude PE
- 1 or more YEARS items → use the lower threshold
That is elegant because it links clinical suspicion with biomarker interpretation.
In bedside terms:
If the patient does not really look like PE, you can tolerate a little more D-dimer noise.
If the patient does look like PE, you become stricter.
That is good emergency medicine.
Pregnancy
Pregnancy is different.
The guideline says pregnancy-adapted YEARS may be reasonable in pregnant patients, and this is one of the most important updates because it helps reduce unnecessary chest imaging in a population where indiscriminate CT use is especially undesirable.
Also important: if the pregnant patient has leg symptoms and compression ultrasound is positive for DVT, you may not need chest imaging to start treatment.
Step 7: High probability means imaging, not more hedging
If the patient is high clinical probability, go to diagnostic imaging.
Do not hide behind D-dimer.
This is a major pitfall in real life. People sometimes order D-dimer because it feels faster, cleaner, or less confrontational than committing to CT. But that is not the right logic.
When the probability is high, the question is no longer:
“Can I cheaply screen this out?”
It becomes:
“Can I confirm or exclude this quickly enough to change outcome?”
That usually means imaging.
Step 8: Choose the right imaging test
CTPA
The guideline continues to favor CT pulmonary angiography as the preferred imaging test in most patients.
Why?
Because CTPA:
- is widely available
- has strong diagnostic performance
- can assess clot burden
- gives RV/LV information
- often identifies alternate diagnoses
That last point matters in the ED.
Sometimes the patient does not have PE — but CT still shows you pneumonia, malignancy, pleural disease, or another explanation.
V/Q scanning
Use V/Q when CTPA is not the right test, especially if contrast is a problem or CT is not feasible.
The guideline favors V/Q SPECT over planar V/Q when available because diagnostic yield is better.
Pregnancy
In pregnant patients with symptoms, suggestive YEARS criteria, and a normal chest x-ray, the guideline says low-radiation-dose CTPA is reasonable over low-dose perfusion scanning. In practice, this is still a center-dependent workflow issue, but the important point is that modern low-dose protocols have changed the old reflexive assumptions.
Step 9: Know what not to use as routine PE diagnostics
This is just as important as knowing what to order.
Echocardiography and Point-of-Care Ultrasound (POCUS)
Echo is not a routine rule-in or rule-out test for PE.
It is useful for:
- unstable patients
- assessing RV strain
- helping with risk stratification
- guiding urgency and escalation
But it should not be used as a stand-alone diagnostic substitute for proper imaging in the stable patient.
Leg ultrasound
Leg duplex is not a routine add-on after a negative CTPA or normal V/Q SPECT just to keep searching for PE. The guideline says that does not add value.
Leg ultrasound is useful when:
- the patient has leg symptoms
- confirming DVT would change management
- in pregnancy or other settings where a positive DVT result may spare chest imaging
That is a focused, purposeful use. Not fishing.
Step 10: Once PE is diagnosed, immediately switch from diagnosis to severity assessment
This is where the 2026 guideline is especially useful.
The old “massive / submassive / low-risk” language is too blunt.
The new guideline introduces AHA/ACC acute PE clinical categories A through E, which better reflect actual physiology and risk.
You do not have to memorize every label to use the concept well.
What matters is that after diagnosis, you immediately assess:
- hemodynamics: BP, MAP, transient hypotension, recurrent hypotension
- perfusion: lactate, urine output, creatinine, mental status
- RV strain: CT or echo
- biomarkers: troponin, BNP
- respiratory Modifier: oxygen need, tachypnea, work of breathing
- clinical risk score: PESI, sPESI, or Hestia
That is the real bedside upgrade.
A PE patient is not just “PE positive.”
He may be:
- low risk and suitable for discharge
- stable but vulnerable
- normotensive but already hypoperfusing
- or on the edge of collapse
so A PE patient is not just “PE positive.”
- Category A (Incidental): They are asymptomatic. The clot was an incidental finding on a scan ordered for something else. They are safe for direct ED discharge.
- Category B (Symptomatic, Low Severity): They have symptoms (like pleuritic pain), but their vitals are normal, and there is no RV strain or biomarker elevation. They are prime candidates for early discharge or a brief observation stay.
- Category C, D, and E: This is where the patient transitions from stable but vulnerable, into pre-shock, and finally into frank cardiovascular collapse.
That distinction changes everything. It gives you a precise roadmap for who goes home and who goes to the ICU.
The practical ER severity frame
After diagnosis, I think in 4 bedside questions:
1. Is the RV suffering?
Look for RV enlargement, septal bowing, reduced TAPSE, RV/LV ratio, biomarker elevation.
2. Is perfusion starting to fail?
Lactate, creatinine, mental status, cool extremities, low MAP even if the systolic number still looks “acceptable.”
3. Is the patient needing more oxygen than the clot burden alone should allow?
The new guideline appropriately gives more weight to respiratory compromise.
4. Is this patient really as “stable” as the blood pressure suggests?
That is a huge point. Normotension does not always mean safety. Some patients are in pre-shock physiology before the cuff fully declares it.
That is a very important update.
Step 11: Start anticoagulation early when appropriate
For confirmed PE, anticoagulation is the foundation unless there is a true contraindication.
The guideline also says that in suspected acute PE with higher-risk features, if imaging is delayed and bleeding risk is low, therapeutic anticoagulation may be beneficial.
That is clinically important.
In real ED practice, the dangerous mistake is not only missing PE.
It is also recognizing likely PE but waiting too long to treat because the scan is delayed.
Of course, this requires judgment. Not every “maybe PE” gets empiric anticoagulation.
But if the patient looks like a real PE, is more than low risk, and imaging is delayed, this is exactly where experienced emergency medicine matters. The 2026 update gives a Class 1 recommendation for Direct Oral Anticoagulants (DOACs) over Vitamin K Antagonists for eligible patients.
What is genuinely updated in this guideline?
A few practical updates stand out.
1. More structured use of D-dimer strategies
The guideline strongly supports age-adjusted D-dimer and supports YEARS to safely reduce imaging in low/intermediate-risk patients.
2. Pregnancy is no longer a diagnostic dead zone
The guideline incorporates pregnancy-adapted YEARS and modern imaging thinking rather than defaulting to vague fear of radiation.
3. A more nuanced severity model
The new AHA/ACC A–E clinical categories are better than the old simplistic bins because they capture patients with respiratory compromise, pre-shock states, and evolving physiology.
4. More emphasis on RV, perfusion, and early deterioration
This is a big one. The guideline does not treat PE as binary. It pushes clinicians to identify who is heading toward trouble even before frank hypotension appears.
5. More explicit role for multidisciplinary care
In patients at increased risk of adverse outcomes, PERT involvement is recommended where available. For the ED, that means earlier coordination, not later rescue.
Pitfalls to avoid
This is the section that actually saves patients.
Pitfall 1: Ordering D-dimer before deciding pretest probability
This is probably the most common error.
A D-dimer without a probability framework is just a machine that generates CT scans.
Pitfall 2: Using PERC in the wrong patient
PERC is for the truly low-risk patient only.
Do not use it in:
- intermediate-risk patients
- high-risk patients
- patients who make you uneasy
- patients with physiology that feels wrong
If your bedside judgment says, “I’m genuinely worried,” you are already past PERC.
Pitfall 3: Using D-dimer to avoid CT in a high-probability patient
That is not evidence-based and not safe.
High pretest probability should move you toward imaging.
Pitfall 4: Treating a positive D-dimer as if it diagnoses PE
It does not.
It only says fibrin turnover is present.
Pitfall 5: Believing normal blood pressure means the patient is fine
Some PE patients are “normotensive” but already showing:
- rising lactate
- worsening oxygen need
- low MAP
- AKI
- RV strain
- altered mentation
- impending fatigue
That is early trouble, not reassurance.
Pitfall 6: Over-relying on clot location and under-reading physiology
A segmental clot in a frail patient with bad cardiopulmonary reserve may matter more than a bigger clot in a younger patient with preserved reserve.
PE severity is not just anatomy.
It is clot burden plus host reserve plus RV response.
Pitfall 7: Using echo as a routine PE exclusion test
Echo is valuable, but not for that.
Use it to understand RV physiology and urgency, not to shortcut the diagnostic process in stable patients.
Pitfall 8: Reflexively adding leg ultrasound after a negative CTPA
The guideline says that is not useful as routine PE diagnostic follow-up.
Pitfall 9: Forgetting pregnancy-specific strategy
Pregnancy changes the pathway. Do not lazily use the generic nonpregnant algorithm.
Pitfall 10: Waiting too long to anticoagulate when the story is strong
If suspected PE is clinically significant, imaging is delayed, and bleeding risk is low, delaying all treatment just because the scan is not back yet can be a mistake.
Pitfall 11: Missing the alternative diagnosis because you anchored on PE
The patient still may have:
- ACS
- tamponade
- pneumonia
- dissection
- pneumothorax
- sepsis
- decompensated heart failure
PE belongs in the differential. It should not erase the differential.
My practical ED approach
This is the bedside script.
First: Does this patient actually deserve a PE workup?
Second: Is the patient stable, or are we already in RV/shock physiology?
Third: What is the honest pretest probability — low, intermediate, or high?
Fourth: If low, can I stop with PERC?
Fifth: If low/intermediate, can D-dimer with YEARS or age-adjustment safely exclude it?
Sixth: If high risk or not excluded, image.
Seventh: Once diagnosed, immediately stage severity using hemodynamics, RV strain, perfusion, biomarkers, oxygen need, and risk score.
Eighth: Start anticoagulation early when appropriate and escalate early if the physiology is worsening.
That is how you evaluate PE like an emergency physician.
Not by memorizing boxes.
By understanding the physiology inside the boxes.
Bottom line
Suspected PE is not mainly a radiology problem. It is a bedside probability-and-physiology problem.
The right approach is:
- think of PE when the story fits
- assess stability first
- estimate pretest probability before ordering tests
- use PERC only in truly low-risk patients
- use D-dimer intelligently, not reflexively
- use YEARS or age-adjusted thresholds to reduce unnecessary imaging
- image high-probability patients
- do not misuse echo or ultrasound
- once PE is diagnosed, switch immediately to severity assessment
- treat early when appropriate, especially if physiology is deteriorating
That is the practical, evidence-based, physiology-based way to do it.